Industries | The Big Take
The Implants Were Supposed to Dissolve. They Didn’t.
Searing pain, pus-filled infections and dying tissue: The BioZorb device tormented patients. Its maker kept complaints from the public.
In the two years following her breast cancer surgery, not a day went by when Mary Munney Griffiths wasn’t in pain. It was different from the burning she felt in her chest during eight weeks of radiation. This was a new sharp, shooting sensation that woke her up at night and stopped her cold in the grocery store.
She worried her cancer had returned, but tests said otherwise. When she finally got a surgeon to operate two years later, the doctor removed 24 plastic shards from her breast.
They were remnants of a medical device called BioZorb, which Griffiths’ physician had implanted to help maintain the shape of her breast. Instead of dissolving within about a year, as intended, it had broken into pieces.

“I’m always worried that there’s more in there,” said the 64-year-old retired administrative assistant in an interview near her home in Syracuse, New York.
Hundreds of patients have had similar experiences – or worse, according to a Bloomberg review of a US Food and Drug Administration database of complaints. They reported searing pain, pus-filled infections and dying breast tissue that they attributed to the device. To alleviate their discomfort, some chose to get mastectomies, losing breasts that — even through radiation and chemotherapy — they might have otherwise kept.
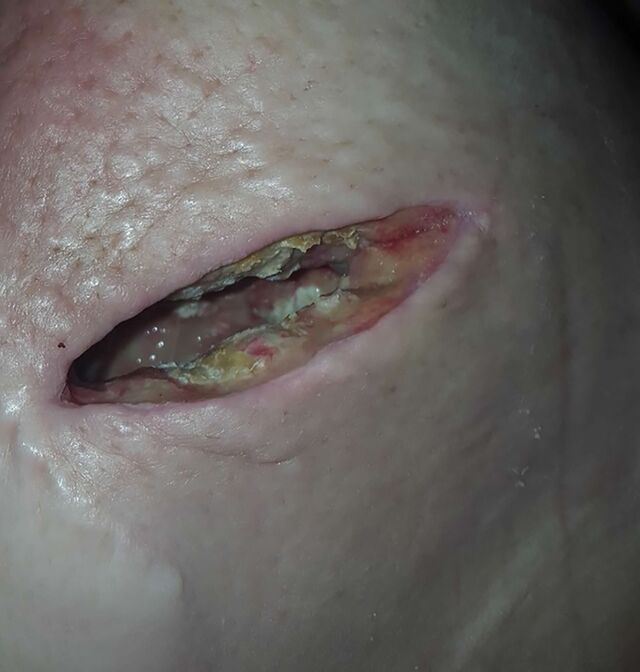
Much of this was kept from the public. Between 2015 and 2022, hundreds of complaints from patients, doctors and others were sent to BioZorb’s makers. The vast majority were only disclosed to the FDA years later, despite a legal requirement that the company submit them within 30 days. Hologic Inc., the device’s maker, received hundreds more complaints after a series of lawsuits starting in 2022.
Hologic didn’t comment on why it didn’t share early reports sooner.
In 2024, the company pulled BioZorb from the market, with the FDA warning it “may cause serious injuries or death.” The agency warned the device caused “adverse events,” including infections, device erosion and other complications, some of which required additional medical treatment. No deaths were reported, it said.
Still, many of the 90,000 breast cancer patients that have had the device implanted continue to live with it in their bodies. And the regulatory system that put it on the market — and kept it there — remains in place.
Cancer Capitalism
This is the second story in a series about how the hunt for profit can harm cancer patients.
“The standards are lax and can be manipulated by expert lawyers and creative manufacturers,” said David Simon, an associate law professor at Northeastern University who co-leads the Amy J. Reed Collaborative for Medical Device Safety. “In a variety of cases, devices are cleared and implanted in humans without ever having been tested on humans.”
Hologic said in an emailed statement that “clinical evidence demonstrates that BioZorb is safe and effective.” The company said it’s committed to meeting the highest standards of patient safety and is working with the FDA to address the agency’s questions. Hologic will face the first trial concerning the device early next year.
Due to the sheer number of people diagnosed annually with breast cancer, treating the disease is a big money-maker. Hologic’s breast health business brings in $1.5 billion annually through a mix of devices and screening and diagnostic tools, like mammography machines.
Medical devices are a particularly appealing revenue stream, in part, because of how easy they are to get to market compared to drugs. The agency doesn’t typically require clinical trials for “moderate risk” devices, a category that includes 99% of all medical devices cleared for sale in the US.
Nearly All Medical Devices Are Approved Without Human Tests
The FDA only requires clinical trials for high-risk devices
This system has existed since 1976, when the FDA created its modern device regulation process. Since then, thousands of products have entered the US market with no human testing. There’s little oversight once they’re widely available, too.
“It’s become the default way to get devices to market because it’s so much easier and less expensive,” Simon said.
In an emailed statement, the FDA said “patient safety is of the highest priority” and that once a device is approved the agency uses “a multifaceted approach” to ensure the safety and effectiveness of devices.
BioZorb was the brainchild of plastic surgeon and inventor Gail Lebovic. Now 68, Lebovic has said her interest in breast cancer began after a family friend died from the disease while she was in medical school at George Washington University in the 1980s. She decided to go into the then-nascent field of oncoplastics, a surgical subspecialty for helping breast-cancer patients maintain the shape of their breasts.
While in training, she started coming up with ideas for new products. Her earliest creations included the MammoPad, a thin cushion meant to make mammograms less painful — which she eventually sold to Hologic for $70 million — and the Expand-a-Band Breast Binder, a bra designed to help with swelling and bruising after surgery.
Around the time she sold MammoPad, she came up with BioZorb: a round plastic coil that doctors could place where they'd removed a tumor. The thinking was that eventually, tissue would grow around the device and the plastic would dissolve. Instead of leaving an empty crater, the breast would heal practically whole.
To get it on the market, she co-founded a company called Focal Therapeutics and started the process of getting FDA clearance. The agency’s 510(k) program governs the sale of devices deemed “moderate-risk.” It doesn’t require clinical trials or lengthy applications. Instead, device-makers have to show their product is similar to something the FDA already approved.
BioZorb’s application listed three “substantially equivalent” products. They were all markers, pieces of metal implanted in people’s breasts that imaging machines can detect to mark where cancer once was.
The device also had a plastic orb-shaped coil that was intended to dissolve as the breast healed. While other medical devices like stents use a similar material, those are made of thin strands that form a netting. In BioZorb’s case, the plastic was as thick as an iPhone cord.
With that application, the FDA allowed BioZorb to come to the US market as a marker in 2012.

When Focal started selling BioZorb it wasn’t just as a marker, according to two people who worked for the company. The device was pitched as something that would help women's breast tissue grow back in a more aesthetically pleasing way before dissolving. Multiple surgeons said they were told it was for cosmetic purposes, too.
The FDA doesn’t allow companies to market their devices for anything other than their approved use.
Lebovic declined to comment on BioZorb’s FDA application or its marketing.
Concerns emerged from some patients and doctors using the device about a year after BioZorb hit the market. Around then, Sarah Blair, a surgical oncologist in San Diego, implanted the device in six patients. Half complained of pain. She said she called Focal to complain.
“They were like, ‘It should dissolve,’” she said about her call with the company. “And I was like, ‘It’s not dissolving.’” She said she couldn’t get the answers she needed, so she stopped using the device.
Lebovic didn’t respond to questions about surgeons who called Focal to complain.
Another surgeon, who asked not to be identified because their employer didn’t authorize them to speak said they were so disturbed after a BioZorb they implanted began poking through a woman’s breast that they stopped using the device. Then, they said they told another surgeon set to operate on a family member who had been diagnosed with breast cancer that there was “no f–king way” they could use BioZorb on her.
For any FDA-approved products, the agency requires companies to submit complaints they receive that “reasonably” could be linked to them. Over BioZorb’s first seven years on the market, Focal received more than 100 reports from patients, doctors and others that rose to that level, according to the FDA’s database. Focal only submitted one of those within 30 days, as required by law.
Lebovic didn’t address questions about the company’s process for sharing complaints with the FDA.
•••
In 2018, Hologic paid $125 million to buy Focal. After the sale, Lebovic told GW’s alumni magazine how gratifying it was to see the device in the world.
“When you become an entrepreneur and invent something that improves patient care, now you can impact millions of women all at once,” she said in the piece.
Soon after the purchase, Hologic started receiving complaints about BioZorb. One woman said she felt like she had bugs crawling under her skin, another said her breast turned black and was leaking blood and fluid. Many said they wished they had never had the device implanted in the first place. The company only sent a handful of the complaints to the FDA over the next few years.
-
Date: Dec. 2022
Complaint:
Post-surgery, my breast turned black (I have fair skin) from a hematoma. After a visit to the emergency room when my breast was leaking blood and fluid, I was diagnosed with a seroma. My surgeon…determined that i was experiencing a foreign body reaction to the Biozorb device, and it was surgically removed [in] 2024.The complications delayed my radiation, caused significant stress, and created additional risk and expense, not only because I needed an additional surgery, but also because the plan to complete my radiation in 2022 was not realized. I therefore had to meet my insurance deductible again in 2023 and pay more out of pocket for radiation. Read the full complaint here.
-
Date: April 2020
Complaint:
Biozorb implant source of constant pain, requiring additional medical follow-up, pain medication and physical therapy. I was told it would absorb in 12 months and now the breast surgeon is saying two years. I will probably need to have the Bbiozorb [sic] removed leading to additional surgery and now facing mastectomy versus lumpectomy. At the very least, I am facing additional reconstructive surgery. Biozorb is such a bad product. How did it ever get approval? Read the full complaint here.
-
Date: June 2018
Complaint:
It felt like there were bugs crawling around under my skin. I developed retraction of the skin and deviation of the nipple. Finally, the surgeon recommended removing the Biozorb device. I opted to have a complete mastectomy as I didn't like the way my breast was now deformed and the radiation would maybe make it worse. As soon as the breast tissue and Biozorb device was removed my skin returned to a normal color and pain free. I wish I had just had the breast removed and done with. I have been under treatment for 14 months and even now I have to have one last reconstruction surgery next week. Read the full complaint here.
-
Date: March 2024
Complaint:
Severe pain a few days after initial op. At post-op (a week after surgery) a hematoma started to develop…was admitted for emergency surgery where they found that the Biozorb had broken in half and likely was stabbing me internally and caused the hematoma. Read the full complaint here.
-
Date: Nov. 2016
Complaint:
I had a partial mastectomy where a Biozorb device was implanted in my breast. It was supposed to be reabsorbed in a year, it has now been close to three years, and it is still not fully absorbed and not only has caused me chronic pain issues. Had I been fully informed of the honest and negative effects of this device I would never have agreed to have this device implanted. It has prevented me from being able to get post-cancer mammograms because of the pain issue. Read the full complaint here.
Meanwhile, doctors kept inserting BioZorb into their patients, including Griffiths.
Griffiths said her surgeon made the decision to get BioZorb an easy one. As she was preparing to have her tumor removed, the doctor said BioZorb would help her keep her breast’s shape. Placing it wouldn’t even require a second surgery: the surgeon could implant it when she removed the cancer.
Griffiths trusted her doctor, and still doesn’t blame her for what happened. “She’s wonderful,” Griffiths said. “We had a very good rapport.”
The pain began immediately. When it didn’t go away after a few months, Griffiths said she asked her doctor to take BioZorb out. She said her physician told her to wait, that it would dissolve within about a year. She couldn’t wear a bra or seatbelt without wincing. Two years later, the surgeon finally relented. Griffiths said even the surgeon couldn’t believe what she ended up pulling out of Griffiths’ chest.
“You’re supposed to trust your doctor,” Griffiths said. But she’s since learned “doctors aren’t gods. They’re getting told things, too — by companies.”
Griffiths’ doctor didn’t respond to requests for comment.
As complaints to the company mounted, Hologic continued to promote BioZorb. In 2022, Nimmi Kapoor, a breast surgical oncologist with UCLA Health, attended a company-sponsored course that promoted BioZorb’s cosmetic benefits. She used it a couple times after that, but abandoned it after determining it wasn’t much better than manipulating breast tissue to get the same effect.

“It’s not unsurprising to me that it didn’t always absorb,” she said in an interview, comparing the device’s material to a plastic toy.
It was only during legal proceedings that Hologic started releasing batches of complaints to the FDA. More than 50 came in a two-day span starting in September 2023. Hundreds more would follow over the next two years.
This influx of reports apparently caught the FDA’s attention. In February 2024, the agency released a “safety communication,” telling doctors to warn patients there might be a risk of serious complications with BioZorb and advising them to monitor those who’ve had it implanted.
Early BioZorb Complaints Took Years to Reach the FDA
Hologic, meanwhile, told some investors not to worry.
Chief Operating Officer Essex Mitchell referred to the issues with BioZorb as “more of an administrative recall,” he said on a July 2024 call with industry analysts. The med-tech firm simply needed to update some of the language on its packaging, he said.
“We are still selling our product, feel great about it, and are working through that,” Mitchell said.
The next day, FDA inspectors showed up at Hologic’s headquarters in Marlborough, Massachusetts, according to a letter the FDA sent Hologic in December. Inspectors combed through the company’s files related to BioZorb. They found Hologic didn’t have sufficient data or “verification testing” to prove the device would dissolve, the letter said. There were other missing pieces too, according to the FDA’s letter, like a lack of evidence the company had properly guided surgeons as to how deeply to implant the device or how to use it on different body types.
Hologic took BioZorb off the market shortly after the visit.
On a call with investors Hologic Chief Financial Officer Karleen Oberton called the impact of BioZorb “de minimis,” she said. “It was a revenue line item that was less than $10 million in ’24.” Hologic’s total sales for its 2024 financial year were more than $4 billion.
The FDA is continuing to monitor the situation. Hologic hasn’t “appropriately evaluated” when the device would be expected to dissolve, how radiation may interact with it and whether it could interfere with detecting any future cancer, according to the FDA’s warning letter. All of this is necessary, the FDA said, to determine whether some women may still need to have the device taken out.
The company told the FDA it has no plans to further study BioZorb, according to the agency’s December letter. A Hologic spokesperson said the company is working with the FDA to address its concerns in a comprehensive and timely manner.

“Their response was like ‘We made a mistake and we’re trying to quickly wipe our hands,’ but it didn’t really show concern for the patients,” said Vinay Rathi, a physician and health policy researcher who has studied medical device regulation at Ohio State University Wexner Medical Center. “It’s like: ‘We can exit this product line, but without surgery, you can’t.’”
Meanwhile, the FDA has received more complaints, bringing the total number to 549 as of Aug. 29.
Getting the shards of BioZorb removed hasn’t solved all of Griffiths’ problems.
Her breast doesn’t look like it used to, the skin where the BioZorb once was has hardened, leaving a large lump behind. “Looking at myself in the mirror, I can’t stand it,” she said.
Her annual mammogram revealed an abnormality where BioZorb used to be. The results of a two-hour biopsy came back negative for cancer, but she fears that the lump left behind by BioZorb — or perhaps a shard of the device — will constantly cause doctors to question whether the cancer has come back.
She’s also still in pain and now considering a mastectomy.